|
Issue |
Our
Solutions |
|
CMS Submission Toolkit |
Toolkit saves time by eliminating
redundancies
Integrated design, implementation and test processes |
|
CMS Report
Requirements |
Standard CMS
submissions - PQRS, GPRO,
eRX, QRDA
Custom Specialist
QCDR reports & metrics
Other standards: CGCAHPS, NQF, HL7 |
|
Locus
of Control |
External: QCDR_HISP cloud
(SaaS)
Internal: Licensed
QCDR_HISP dedicated server
(QHP) |
|
Automated Review |
Data type, format & boundary testing
Schema Validation
using CST |
|
Adaptable Data Import |
Configurable Data Crosswalk
CCDA, QRDA, Custom XML & Flat files |
|
Datacomm Flexibility |
Standard SECURE datacomm interfaces
DSM , S/MIME,XDR/XDM, NwHIN, XDS, FTPS |
|
Zero
Trust Security |
HIPAA Compliance (2013)
Triple DES encryption |
> Our QCDR Private Cloud
providers HIPAA secure electronic publishing services for CMS
Submissions. More...
> See details on our CMS Submission
Electronic Publishing.
More..."
> We merge multiple data sources specific to
your metrics.
More...
> We
save your time with our CST (CMS Submission Template)
> The CST
provide
a detailed schema and
methods for automated review at discrete stages of the report generation
process to meet the stringent CMS data requirements.
More...
> We use valuable clinician time
judiciously by isolating elements of the CMS electronic report the require
clinical judgment. More...
> Our
HIPAA datacomm securely exchanges
clinical data and submissions. More...
CMS Submission process - without
QCDR-HISP.ORG:
You will find precious clinician time wasted on re-inventing design,
implementation and the intensive review processes for the massive
amounts of clinical data inherent in CMS submissions.
CMS Submission process - with
QCDR-HISP.ORG:
Your expedited
QCDR submissions will meet tight CMS deadlines and be applied to
assess and plan improvement cycles of your relevant healthcare
processes.
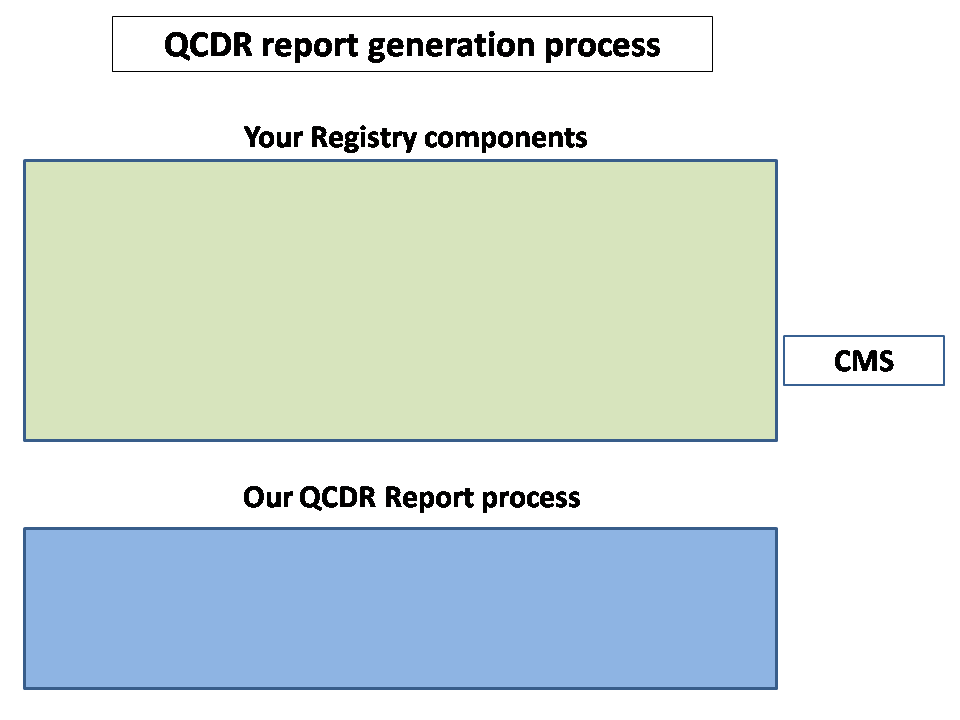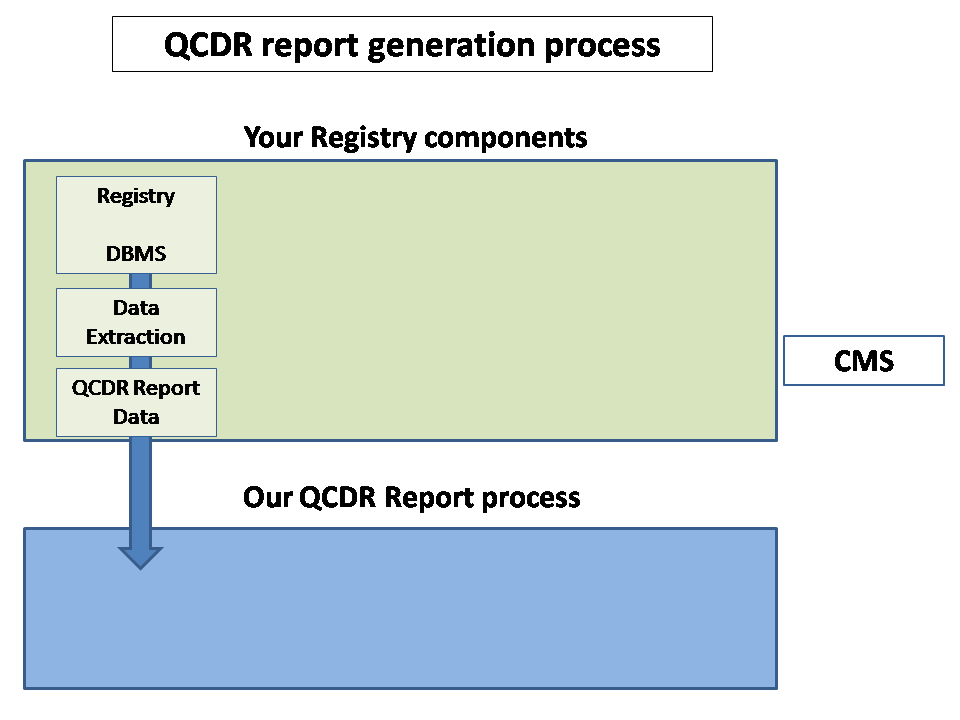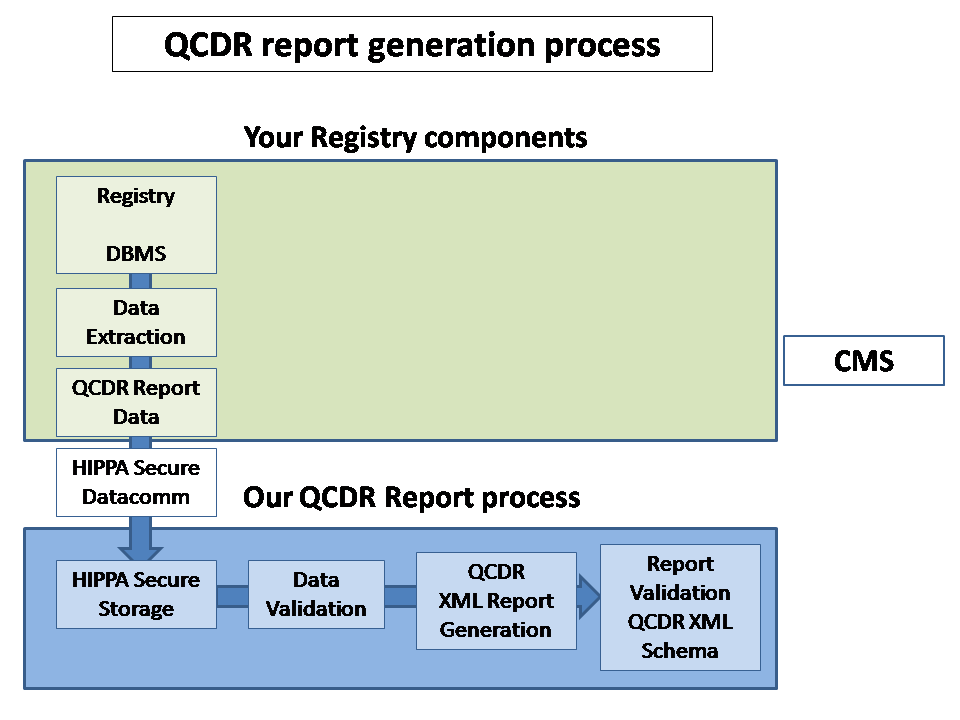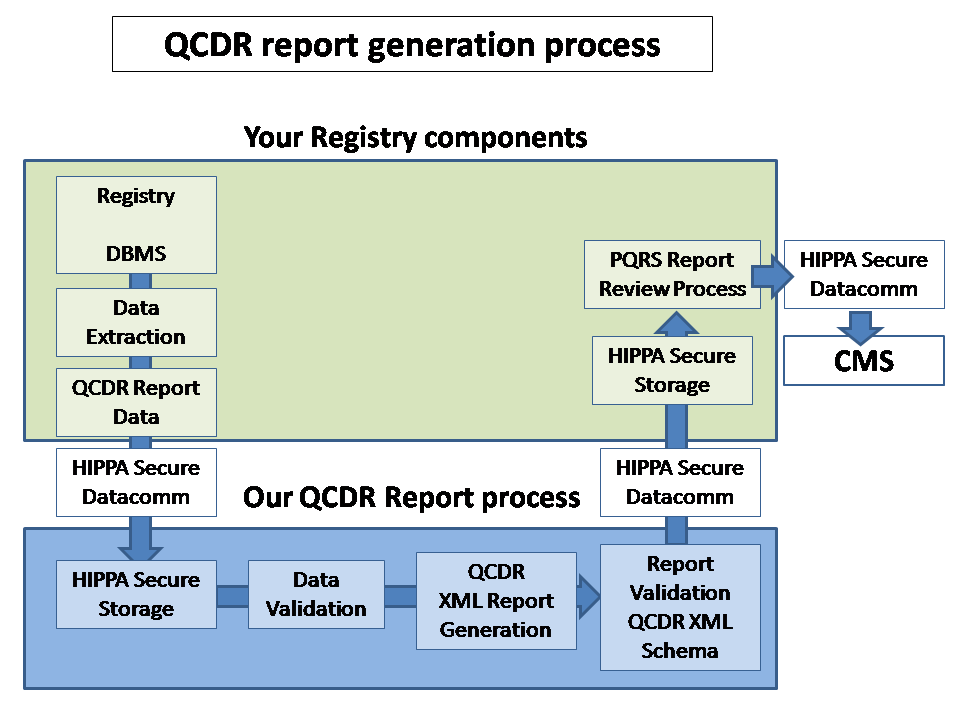 For higher resolution, click
here
For higher resolution, click
here